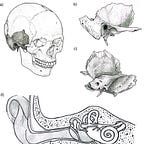
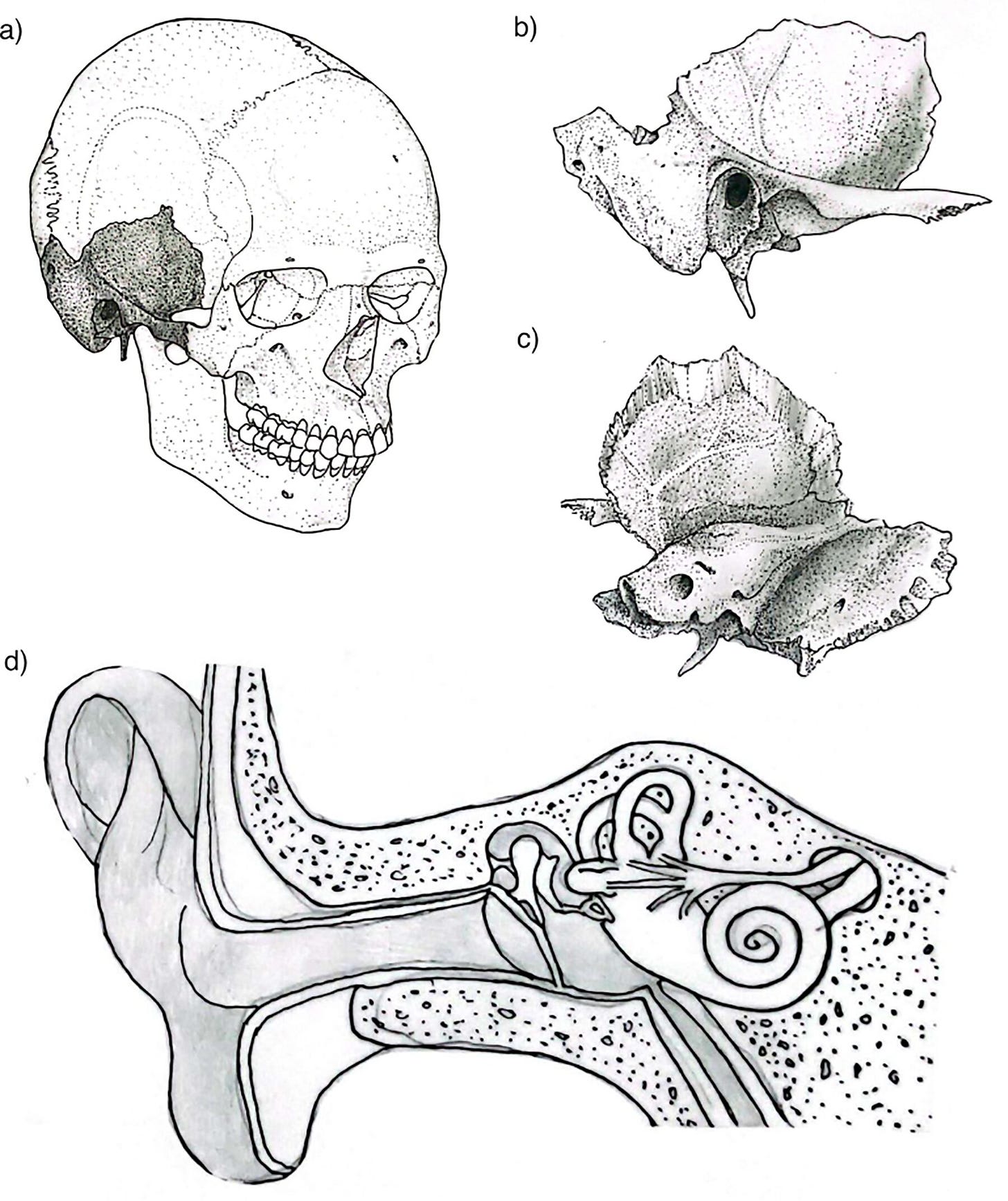
The petrous bone is not beautiful. It is a dense, pyramidal wedge buried in the base of the skull, housing the cochlea and semicircular canals, its name taken from the Latin petrosus, meaning rock-like. It weighs almost nothing. You could balance one on your fingertip. Under most circumstances it would register as unremarkable.
In paleogenomics, it has become the most fought-over fragment of bone on earth.
The cochlea within the petrous is the densest, most highly mineralized structure in the human skeleton. It finishes forming by the eighth week of fetal development and then, essentially, stops changing. Unlike most bone, it doesn’t remodel after birth. It sits there, stable, shielded, preserving its chemistry for centuries or millennia after death. When researchers began testing which skeletal structures yield the highest-quality ancient DNA, the cochlea came out on top, beating out teeth, femora, and everything else. The petrous became the default target for nearly every major paleogenomics lab in the world.
According to metadata compiled in the Allen Ancient DNA Resource (AADR v9), a curated database of ancient human genomes, ancient DNA has been retrieved from the petrous bone in 47% of all sequenced individuals. In 96% of those cases, it was the only bone sampled.
That is a lot of irreversible drilling.
Petrous Fever
The phrase “petrous fever” appears in a 2024 paper in Current Anthropology,1 describing the gap between what ancient DNA labs claim to prioritize and what they actually do in practice. It describes a pattern of extensive, often redundant sampling from skeletal collections, frequently without clearly defined research questions, driven by the logic of data generation rather than specific inquiry. More than 10,000 ancient human genomes have been generated over the past decade, the majority relying on petrous bones. The momentum is real. So is the problem it creates.
The cochlea is not just a DNA vault. The bony labyrinth inside the petrous bone is also a morphological record. Its shape reflects population history and has been used to trace migration and evolutionary divergence across time. The cranial base, of which the petrous is a component, carries a strong phylogenetic signal less susceptible to environmental noise than other cranial regions. And because the petrous forms so early in development and remains so stable, its fine structural details survive taphonomic processes that destroy nearly everything else. Studies of inner ear anatomy, basicranial morphology, stable isotopes, and radiocarbon dates have all increasingly relied on the same small piece of bone.
When you drill into the cochlea to extract DNA, you destroy it. The morphological data, the histological information, the stable isotope signal, the inner ear geometry that might have told you something about population dispersal or evolutionary change: all of it is gone. Researchers visiting skeletal collections have encountered cases where both petrous bones from a single individual were removed entirely, one for genetic analysis and one for radiocarbon dating, sometimes both for separate DNA extraction attempts by the same lab, even when a single sample would likely have been sufficient.
The question2 that Lumila Paula Menéndez and colleagues at the University of Bonn, along with a large international team, set out to address is both practical and ethical: can you digitally preserve a petrous bone before destroying it for molecular analysis, and if so, does the imaging process itself damage the DNA you’re trying to extract?
The Worry About X-Rays
Micro-computed tomography, or µCT, works by passing X-rays through a sample from multiple angles and using algorithms to reconstruct high-resolution three-dimensional cross-sections. For the inner ear, it produces extraordinarily detailed models of the cochlea and semicircular canals, images that can be archived, shared, and analyzed without any physical contact with the original bone. Its advocates have been pushing for it to become a standard step in any research workflow involving the petrous.
The hesitation came from a reasonable concern. CT scanning uses ionizing radiation, and ionizing radiation can damage DNA. If scanning compromises the very molecules researchers are trying to recover, the approach defeats its own purpose.
Prior work on animal samples and other human bones had suggested that standard µCT parameters fall below radiation doses likely to cause meaningful DNA degradation. But the petrous bone is not a generic skeletal element. Its microstructure and diagenetic history are distinctive. Its density means it interacts with radiation differently. And because it yields such unusually high proportions of endogenous ancient DNA, even minor damage could have disproportionate effects. No previous study had tested µCT effects directly on archaeological human petrous bones.
Menéndez and her team found themselves in a position to do exactly that, not by design but by circumstance. In 2021, Menéndez contacted a lead geneticist who was already sampling petrous bones from South American archaeological individuals for a population genomics project. She proposed adding µCT scans before any destructive procedures. Some samples were scanned. Others, already in the pipeline, were not. The mixed dataset created a rare opportunity for comparison.
The team analyzed 93 petrous bone samples from Middle and Late Holocene archaeological contexts across the Andes, Central Argentina, and the Cuyo region, spanning roughly 6,000 to 200 years before present. Fifty had been µCT-scanned before molecular analysis; 43 had not. They compared six standard parameters used to evaluate ancient DNA quality and authenticity: endogenous DNA content, read length, cytosine deamination patterns, which are a chemical hallmark that confirms the ancient origin of DNA sequences, and contamination estimates based on both mitochondrial and nuclear DNA. The scanning itself was carried out on a Skyscan 1176 Bruker scanner at the Faculty of Medicine, Paris Diderot University, at 35 µm voxel size, 65 kV, and with a 0.5 mm aluminum filter to reduce unnecessary radiation exposure.
No statistically significant differences emerged between the scanned and unscanned groups across any of the six parameters. Endogenous content was similar. Read length was similar. Deamination patterns indicating authentic ancient origin were nearly identical between groups. Nuclear contamination estimates showed no difference at all.
One parameter came close to significance. Mitochondrial contamination estimates were marginally higher in the scanned samples, with a p-value of 0.051. But 48 of 49 scanned samples remained below the standard 5% threshold for genomic analysis, and the team’s most parsimonious explanation is selection bias: researchers tend to choose visually well-preserved, more complete bones for scanning, which may incidentally have had more handling and thus slightly more exposure to environmental contaminants. No corresponding effect appeared in nuclear contamination, which would be expected if radiation-induced damage were the mechanism. And it is worth noting that the highest endogenous DNA content in the entire dataset was found in the scanned group, an outcome that makes no sense if scanning is degrading the material.
The absence of a detectable effect under these specific conditions is not proof that no effect exists under all conditions. The authors are careful here. Synchrotron scanners, which can deliver far more intense radiation, are a separate matter. The study’s design, because it wasn’t a matched-pair experiment, limits the ability to attribute every observed pattern solely to the scanning variable. Subtle effects at the margins of detectability might still exist. Future controlled experiments using matched samples from the same individuals would strengthen the picture considerably.
There is also one caveat that sits slightly outside the scope of this study but matters for field practice: some evidence suggests that µCT scanning can reduce collagen yield, which could affect the reliability of radiocarbon dates from bone. Researchers building multi-method workflows that include 14C analysis should factor this in.
What Happens When You Slow Down
The paper’s practical argument follows from its result. If µCT scanning under standard parameters doesn’t compromise ancient DNA, there’s no technical reason to skip it before destructive sampling. Which means the barrier that many labs cited for not scanning first is substantially weaker than it seemed. What remains are the logistical and financial barriers, and the cultural inertia of a field that has organized itself around rapid data production.
The workflow Menéndez and her team propose is sequential and deliberately unhurried. Before any bone powder is collected, before any drilling begins, the petrous goes through macroscopic assessment, biological profiling and osteobiographical analysis, digital preservation via CT and surface scanning, and compositional pre-screening using techniques like X-ray fluorescence and near-infrared spectroscopy to evaluate collagen preservation and guide sample selection. Molecular sampling, with a clear research question in hand, comes last.
This is not a radical reorganization. It is a sequencing change. It asks researchers to wait a few steps before drilling, to ask what other questions might be answered by this bone before it’s gone.
The obstacles are real. CT scanners are not uniformly accessible. The global average cost of a scan is around $100, and many research groups operating with limited budgets have not planned for imaging. Some institutions house their own µCT equipment with trained operators. Others operate in regions where even medical CT scanners are unavailable within thousands of kilometers. The paper addresses this and suggests collaborations with public medical facilities, universities, and private medical centers as partial solutions. Portable surface scanning and photogrammetry present fewer logistical barriers and can often be done in situ.
The geopolitical dimension here is not incidental. Most of the ancient human samples driving the aDNA boom originate in the Global South. Most of the large, well-resourced laboratories generating genomes from them are in the Global North. Menéndez and her colleagues are explicit about this: labs in core research countries have both the capacity and a particular responsibility to lead the transition toward more sustainable practices, precisely because they are the primary beneficiaries of destructive sampling from collections held in institutions that often lack the infrastructure to implement better practices on their own.
The proposed workflow is also an argument about what counts as evidence. The petrous bone that tells you about genome-wide ancestry might also have told you something about inner ear geometry and population dispersal. About diet and mobility through stable isotopes. About developmental biology. Once it’s powder, none of those conversations are possible. The field has been systematically foreclosing its own future questions.
Researchers at collections across multiple continents have encountered individuals from whom both petrous bones were removed entirely, nothing left. It’s difficult to know how many of those samples were digitally preserved before the drilling started, the paper notes. Probably only a small fraction.
The proposed five-step workflow won’t be adopted uniformly or quickly. There are labs that will keep sampling as they always have, and there are collections that will continue to be depleted without the kind of careful sequencing this paper advocates. But the technical argument for delay has weakened. The concern about X-ray damage to DNA, under standard µCT conditions, appears to be manageable. What’s left is a choice about what kind of science gets done with what remains.
Further Reading
Mallick S, Micco A, Mah M, et al. The Allen Ancient DNA Resource (AADR): a curated compendium of ancient human genomes. Scientific Data 11(1):182 (2024). https://doi.org/10.1038/s41597-024-03031-7
Pinhasi R, Fernandes D, Sirak K, et al. Optimal ancient DNA yields from the inner ear part of the human petrous bone. PLoS One 10(6):e0129102 (2015). https://doi.org/10.1371/journal.pone.0129102
Immel A, Le Cabec A, Bonazzi M, et al. Effect of X-ray irradiation on ancient DNA in sub-fossil bones: guidelines for safe X-ray imaging. Scientific Reports 6:32969 (2016). https://doi.org/10.1038/srep32969
Duval M, Martín-Francés L, Wood R. On the impact of micro-CT scanning on radiocarbon dating of fossil material: a cautionary note for the palaeoanthropological community and beyond. Radiocarbon 67(4):709–718 (2025). https://doi.org/10.1017/rdc.2025.23
Sirak K, Fernandes DM, Cheronet O, et al. A minimally-invasive method for sampling human petrous bones from the cranial base for ancient DNA analysis. BioTechniques 62(6):283–289 (2017). https://doi.org/10.2144/000114558
Källén A, Mulcare C, Nyblom A, Strand D. Petrous fever: the gap between ideal and actual practice in ancient DNA research. Current Anthropology 65(6):990–1009 (2024). https://doi.org/10.1086/733328
Menéndez, L. P., Luisi, P., López-Sosa, M. C., Da Silva, S. M., Buck, L. T., Kuzminsky, S. C., Sánchez, M. Q., Le Maître, A., Chappard, C., Rios, C., Groh, H., Siles, W., Hernández, G. M., Becerra-Valdivia, L., Argüelles, J. M., Yáñez, B., de la Fuente Castro, C., Tamburrini, C., Garcia-Deister, V., … Rascovan, N. (2026). µCT scanning effects on aDNA and a multi-step workflow for archaeological petrous portions. PloS One, 21(4), e0334682. https://doi.org/10.1371/journal.pone.0334682